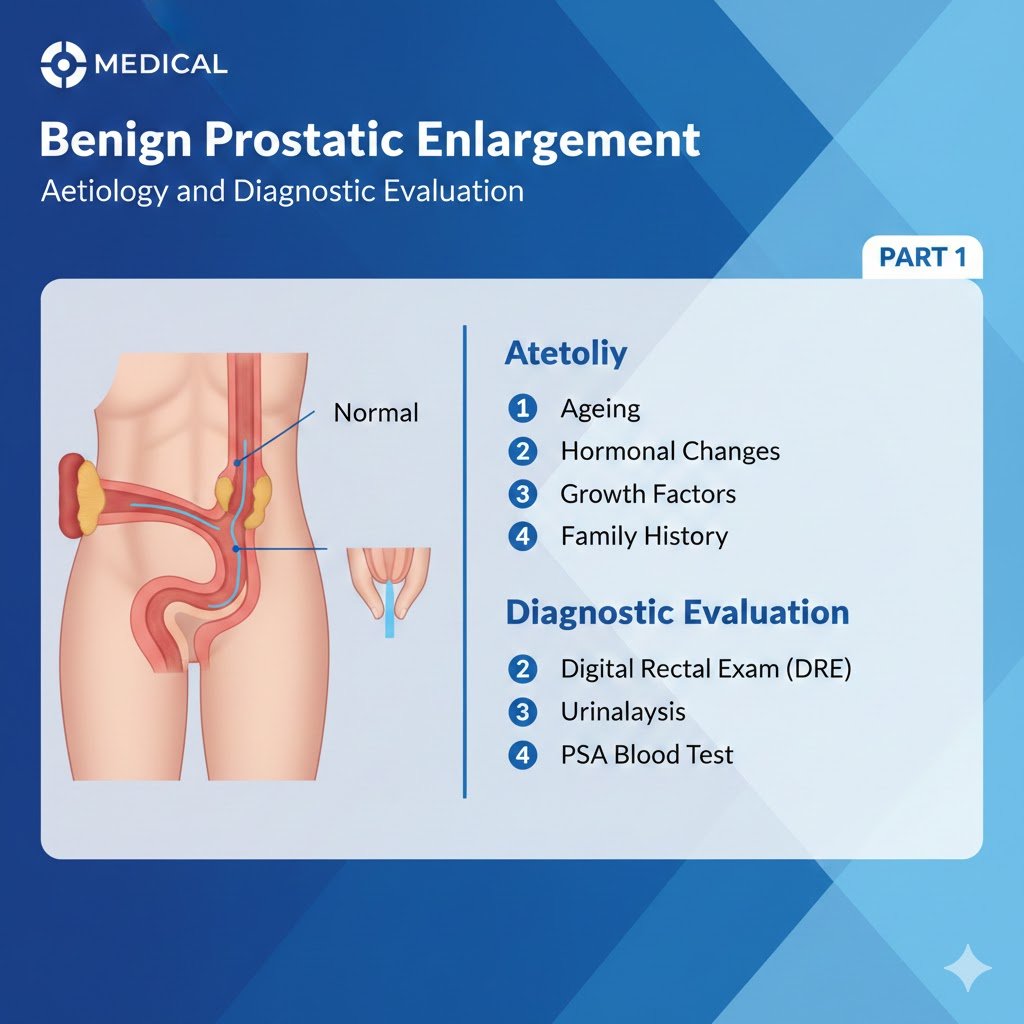
Benign Prostatic Enlargement
Benign Prostatic Enlargement Pharmacological treatment, plant Extracts-phytotherapy PART 2 Comprehensive Review Article Prof. Dr. Semir. A. Salim. Al Samarrai As conservative treatment, the watchful waiting strategy (WWS) is a viable option for benign prostatic enlargement disease management. Many men with LUTS are not troubled enough by their symptoms to need drug treatment or surgical intervention. All men with LUTS should be formally assessed prior to any allocation of treatment in order to establish symptom severity and to differentiate between men with uncomplicated (the majority) and complicated LUTS. Watchful waiting strategy (WWS) is a viable option for many men with non-bothersome LUTS as few will progress to AUR and complications (e.g. renal insufficiency or stones) [1,2], whilst others can remain stable for years [3]. In one study, approximately 85% of men with mild LUTS were stable on Watchful waiting strategy (WWS) at one year [4]. A study comparing Watchful waiting strategy (WWS) and transurethral resection of the prostate (TURP) in men with moderate LUTS showed the surgical group had improved bladder function (flow rates and PVR volumes), especially in those with high levels of bother; 36% of Watchful waiting strategy (WWS) patients crossed over to surgery within five years, leaving 64% doing well in the Watchful waiting strategy (WWS) group [5,6]. Increasing symptom bother and PVR volumes are the strongest predictors of Watchful waiting strategy (WWS) failure. Men with mild-to-moderate uncomplicated LUTS who are not too troubled by their symptoms are suitable for Watchful waiting strategy (WWS). The behavioral and dietary modification for benign prostatic enlargement disease management is customary for this type of management to include the following components: • education (about the patient’s condition); • reassurance (that cancer is not a cause of the urinary symptoms); • periodic monitoring; • lifestyle advice [3,4,7,8] such as: reduction of fluid intake at specific times aimed at reducing urinary frequency when most inconvenient (e.g., at night or when going out in public); avoidance/moderation of intake of caffeine or alcohol, which may have a diuretic and irritant effect, thereby increasing fluid output and enhancing frequency, urgency and nocturia; use of relaxed and double-voiding techniques; urethral milking to prevent post-micturition dribble; distraction techniques such as penile squeeze, breathing exercises, perineal pressure, and mental tricks to take the mind off the bladder and toilet, to help control OAB symptoms; bladder retraining that encourages men to hold on when they have urgency to increase their bladder capacity and the time between voids; reviewing the medication and optimising the time of administration or substituting drugs for others that have fewer urinary effects (these recommendations apply especially to diuretics); providing necessary assistance when there is impairment of dexterity, mobility, or mental state; treatment of constipation. Evidence exists that self-management as part of Watchful waiting strategy (WWS) reduces both symptoms and progression [7,8]. Men randomised to three self-care management sessions in addition to standard care had better symptom improvement and QoL than men treated with standard care only, for up to a year [7]. A SR and meta-analysis found reasonable certainty in estimates that self-management intervention significantly reduced symptom severity in terms of IPSS at six months compared with usual care [9]. The reduction in IPSS score with self-management was similar to that achieved with drug therapy at six to twelve weeks. Self-management had a smaller, additional benefit at six weeks when added to drug therapy [9]. The Plant Extracts- phytotherapy with herbal medicinal products and their drug preparations are made of roots, seeds, pollen, bark, or fruits. There are single plant preparations (mono-preparations) and preparations combining two or more plants in one pill (combination preparations) [10]. Possible relevant compounds include phytosterols, ß-sitosterol, fatty acids, and lectins [10]. In vitro, plant extracts can have anti-inflammatory, anti-androgenic and oestrogenic effects; decrease sexual hormone binding globulin; inhibit aromatase, lipoxygenase, growth factor-stimulated proliferation of prostatic cells, α-adrenoceptors, 5 α-reductase, muscarinic cholinoceptors, dihydropyridine receptors and vanilloid receptors; and neutralise free radicals [10-12]. The in vivo effects of these compounds are uncertain, and the precise mechanisms of plant extracts remain unclear. The extracts of the same plant produced by different companies do not necessarily have the same biological or clinical effects; therefore, the effects of one brand cannot be extrapolated to others [13]. In addition, batches from the same producer may contain different concentrations of active ingredients [14]. A review of recent extraction techniques and their impact on the composition/biological activity of available Serenoa repens based products showed that results from different clinical trials must be compared strictly according to the same validated extraction technique and/or content of active compounds [15], as the pharmacokinetic properties of the different preparations can vary significantly. Heterogeneity and a limited regulatory framework characterise the current status of phytotherapeutic agents. The European Medicines Agency (EMA) has developed the Committee on Herbal Medicinal Products (HMPC). European Union (EU) herbal monographs contain the HMPC’s scientific opinion on safety and efficacy data about herbal substances and their preparations intended for medicinal use. The HMPC evaluates all available information, including non-clinical and clinical data, whilst also documenting long-standing use and experience in the EU. European Union monographs are divided into two sections: a) Well established use (marketing authorisation): when an active ingredient of a medicine has been used for more than ten years and its efficacy and safety have been well established (including a review of the relevant literature); and b) Traditional use (simplified registration): for herbal medicinal products which do not fulfil the requirements for a marketing authorisation, but there is sufficient safety data and plausible efficacy on the basis of long-standing use and experience. Table 1 lists the available EU monographs for herbal medicinal products and the current calls for update. Table 1: European Union monographs for herbal medicinal products Only hexane extracted Serenoa repens (HESr) has been recommended for wellestablished use by the HMPC. Based on this a detailed scoping search covering the timeframe between the search cut-off date of the EU monograph and May 2021 was conducted for HESr. A large meta-analysis of 30 RCTs with 5,222 men
Benign Prostatic Enlargement قراءة المزيد »